Home » Spotlight » Memorial first to switch to single-use duodenoscopes
Memorial first to switch to single-use duodenoscopes
https://www.whatjobs.com/news/what-news-topical/memorial-first-to-switch-to-single-use-duodenoscopes

By Nagasunder in Spotlight, posted October 4, 2021
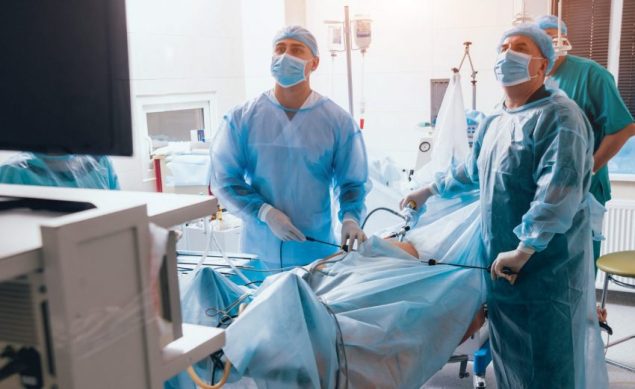
Memorial Health System is the first hospital jobs in the United States to use an Ambu USA sterile, single-use duodenoscope for endoscopic retrograde cholangiopancreatography treatments.
Dr. Juan Tejada, a gastroenterologist and advanced endoscopist at Memorial Health System, was the state's first–and one of the country's first–to employ the technology.
Duodenoscopes are used to visually check the duodenum, the first segment of the small intestine immediately beyond the stomach, in an estimated 600,000 procedures in the United States each year. The devices are essential in diagnosing and treating illnesses such as gallstones, pancreatitis, tumors, liver problems, and cancer of the bile ducts or pancreas.
Duodenoscopes are also utilized in biliary interventions, sphincterotomies, stone removal, and the implantation of expandable metal biliary stents.
Even with the most severe cleaning protocols, studies reveal that reusable duodenoscopes can cause cross-contamination among patients. The US Food and Drug Administration(FDA) proposed in 2020 that endoscope manufacturers shift to new and novel duodenoscope designs, including single-use scopes and components, to aid enhance cleaning of the complicated equipment and prevent cross-contamination between patients. That danger increased after COVID-19 raised awareness about patient safety measures.
“Part of the art of saving lives while using technology is to know when the right time is to change to something simpler while continuing to keep in mind the human being behind the process. I believe that single-use duodenoscopes move us in this direction,” said Dr. Juan Tejada.
In July 2020, the FDA gave the Ambu aScope Duodeno 510(k) clearance and Breakthrough Device Designation, which is given to innovative medical devices that have the potential to deliver more effective treatment or diagnosis of life-threatening or irreversibly debilitating diseases or conditions.
Ambu's aScope Duodeno eliminates the need for reprocessing and repairs in reusable duodenoscopes. It provides high-definition imaging of mucosa and anatomical structures in the upper gastrointestinal system, a shorter turnaround time to accommodate more patients, precise positioning, and a 130-degree wide-field view. The aScope Duodeno also eliminates the need for sterilizing gases and liquid chemical germicides for high-level disinfection, both of which can influence air and water quality.
“At Memorial Health System, our patients are our top priority and the reason we are committed to using the latest technology to help make their experiences as efficient and safe as possible,” said Bob Williams, director of surgical services. “We are proud to have been one of the first in the country to utilize this new, state-of-the-art technology that reduces the risk of infection and allows our doctors and patients to focus on diagnosis and treatment.”
Tejada called the transition “a win for both physicians and patients. “It allows us to advance patient safety through innovative design and provide the safest and best possible outcomes to our patients,” he said.
Source: Marietta Times